Volume : 1 | Issue : 1
Research
Antibacterial and antiviral effect of essential oils combined with non-thermal disinfection technologies for ready-to-eat Romaine lettuce
Birmpa Α,1 Constantinou P,1 Dedes C,1 Bellou Μ,1 Sazakli E,2 Leotsinidis M,2 Vantarakis A1
1Environmental Microbiology Unit, Department of Public Health, University of Rio, Patras, GR 26500, Greece
2Department of Public Health, University of Patras, Rio, Patras, GR 26500, Greece
Received: February 05, 2018 | Published: March 21, 2018
Abstract
The consumer’s demands towards fresh, convenient and safe foods have prompted research into alternative food disinfection technologies. The ability of bacteria and viruses to persist in fresh produce, as well as their low infectious dose, could lead to serious foodborne problems. Our study focuses on both bacterial and viral inactivation by different essential oils (lavender, mint, sage, basil) and by combining alternative non-thermal disinfection technologies (Ultrasound and Ultraviolet Light) with essential oils on fresh ready-to-eat lettuce. The results of the present study showed that when combined UV/Essential Oils were used, enhanced disinfection efficiency was observed, compared to EOs alone (p<0.05) and combined Ultrasound/EOs treatments (p<0.05) for all bacteria. Moreover, significant results (p<0.05) were achieved with Basil and Mint EOs followed by UV and US, compared to the rest EOs for virus disinfection. Results were compared by an analysis of variance followed by Tukey's method with a significance level of p<0.05.
Keywords: essential oils, antibacterial, antiviral, ready-to-eat food matrices, non thermal technologies
Introduction
The important growth in the ready-to-use (RTU) food industry has mainly been attributed to the increasing demand for fresh, healthy and convenient foods. Vegetables and fruits are rich in vitamins and minerals.1 The recommended fruit and vegetable consumption is higher than 400 g/day and consumers should be encouraged to eat at least five servings of fruit and vegetables each day.2,3
Food-borne pathogens are major contaminant of fresh produce and minimally processed ready-to-eat foods.4 Pathogens such as Listeria monocytogenes, Salmonella spp, Staphylococcus aureus and E.coli O156:H7 may cause foodborne illnesses and are present in minimally processed fruits and vegetables.4,5
Consumer demands are to minimize or exclude chemically synthesized antimicrobial and use only natural antimicrobials for food preservation, such as essential oils (EOs).6 EOs are considered Generally Regarded as Safe (GRAS) and are aromatic oily liquids extracted from various plant material as flowers, fruits, herbs, leaves, roots, seeds, and stem.7 EOs have been shown to exhibit antibacterial effects against pathogens and spoilage bacteria.7–10
Essential oils have been investigated as natural sanitizers to control foodborne pathogens as well as the spoilage bacteria which are often detected in minimally processed.11,12 Many in vitro studies have checked the antimicrobial activity of oregano (Origanum vulgare), thyme (Thymus vulgaris) against a variety of Gram-positive, Gram-negative bacteria, yeasts and moulds.7,13 However, there are very limited studies that investigate the antimicrobial efficacy of these natural antimicrobials alone or in combinations with other treatments on vegetable produce.14–16 Sage (Salvia officinalis L.) has been found to offer a range of medical effects. For example, antidiabetic, antihydrotic, antiseptic, cardiovascular, anticancer, antimutagenic, antioxidant, antimicrobial and antiviral.17–20 These natural disinfectants could play an important role in minimizing the spread of pathogenic microorganisms, thus reducing the use of synthetic chemicals which burden further the environment.17 Another common essential oil “basil” (Ocimum basilicum L.; O. basilicum) is a member of the Lamiaceae family, it is an annual herb which grows in many countries.21 Recently the potential uses of O. basilicum essential oil, particularly as antimicrobial and antioxidant agents have also been investigated.22–24
The genus Lavandula (lavender) is comprised of about 25–30 species of flowering plants in the Lamiaceae. (Labiatae) family25 is a well-known essential oil about its antimicrobial26 and antifungal27 properties.
“Mentha piperita” is another essential oil which has high antimicrobial activity due to the presence of its components which are a-terpinene, isomenthone, trans-carveol, pipertitinone oxide and b-caryophyllene. It’s in vitro antimicrobial activity has been studied against opportunistic pathogens such as Candida albicans, Escherichia coli and Staphylococcus aureus.28
In the recent years non-thermal disinfection technologies have widely been used in fresh produce disinfection.29 They have been applied for the decontamination of a wide variety of food products. In advance, essential oils derived from the natural sources have showed broad-spectrum activity, including antifungal, antibacterial and antiviral activities.
Ultrasound technology has been used as an alternative technology to kill bacteria. Recently, it has been proposed the use of ultrasound in combination with other decontamination technologies in order to obtain better disinfection results.30 Combination of ultrasound with essential oils (EOs) is a recent decontamination method which has not been widely studied yet.
In addition, UV-C (190–280 nm) light31 has been used as antimicrobial technology as induces DNA damages, leading to cell death.32 The combined effect of UV-C and essential oils in fresh produce disinfection has not been studied extensively. Tawema et al.33 have studied the above combined effect against two pathogens on fresh cut cauliflower.
The purpose of the present study was to evaluate the antimicrobial and antiviral effect of EOs (lavender, sage, mint and basil) separately and in combination with non-thermal technologies ultrasound or UV on the survival of Escherichia coli, Staphylococcus aureus, Salmonella Enteritidis and Listeria innocua and Adenovirus (hAdV35) into fresh-cut lettuce. According to author’s knowledge this is one of the very few studies that evaluated the antiviral effect of EOs and the first study that investigated the combined antimicrobial and antiviral effect of non-thermal technologies and different EOs.
Material and methods
Food samples
Romaine lettuce (Lactuca sativa L. var. longifolia) was selected to study the level of decontamination with different essential oils. It was selected as it is a commonly consumed vegetable in Mediterranean countries diet and it also ensures an adequate intake of vitamins and antioxidants. Commercially available romaine lettuce was purchased from a local supermarket (Patras, Greece) the day of the experiment and stored under refrigerated conditions (4°C) for 1 h until the time of the experiment.
Bacterial strains
Experiments were conducted using the following bacterial strains: E. coli NCTC 9001, S. aureus NCTC 6571, S. Enteritidis NCTC 6676 and L.innocua NCTC 11288 (HPA, Colingdale, U.K.). Lenticule discs with the microorganisms were rehydrated in 9 mL of peptone saline (0.1%) (CM 0733, Oxoid, U.K.) and after 20 min, working cultures were streaked onto Tryptic Soy Agar (TSA; Oxoid, U.K.), incubated at 37°C for 24 h, and stored at 4°C. Lenticule discs were used in this study as they contain viable microorganisms in a certified quantity (generally accredited according ISO/IEC 17025), produced under reproducible conditions compliant with ISO Guide 34:2009 using authenticated strains from NCTC, NCPF and CECT.
Virus cell lines
HAdV serotype 35 stocks were used according to Birmpa et al., 2016. The initial concentrations of hAdV stock suspensions were quantified by QPCR (Section 2.13) and were calculated to be 108-109 genomic equivalents/ml (GEs/ml).
Culture preparation
Each bacterial type was cultured in 40 mL Tryptone Soya Broth (TSB; Merck, U.K.) at 37°C for 17 h, harvested by centrifugation at 4000×g for 20 min at 4°C and washed three times with buffered peptone water (BPW; Oxoid). The final pellets were resuspended in BPW, corresponding to approximately 106–107 CFU/mL. Bacterial cocktails were prepared by mixing equal volumes (10mL) of each of the tested bacterial type.
Sample inoculation
All vegetables were rinsed with sterile water to remove some of the natural flora or any other matter before treatment. The outer leaves were discarded and the intact internal leaves were removed and weighted to give samples of 10 g. A spot-inoculation method was used to inoculate the pathogenic bacteria on lettuce leaves.32 Likewise, deionized sterile water was spotted on the surface of control samples. To allow bacterial attachment, the samples were air dried on sterile aluminum foil in a class II biosafety cabinet (Cytair 155, FluFrance) for 2 h in 25°C prior to treatments.
For hAdVs the same method was applied to inoculate the hAdVs on the ready-to-eat food products. Briefly, 100 μL (10 drops) of Adeno-35, corresponding to a concentration of 108–109 GEs/ml was spotted with a micropipette on 10 different areas of the surface of each produce. After spiking, the samples with inoculum were dried in a class II biosafety cabinet (Cytair 155, FluFrance), for 20 min at 22±2°C, to allow viral attachments, prior to treatments.
Essential Oils
The essential oils used were lavender (Lavandula hybrida), mint (Mentha piperita), and salvia (Salvia fruticosa) that were obtained from a Greek essential oil company “Myrtis” (http://myrtisfytoria.com/). This supplier extracts essential oils on an industrial scale by steam distillation. All the above plants were cultured in Greece and the essential oils produced in water as a broth. The basil essential oil (Basil oil, methyl chavicol type) obtained from Sigma-Aldrich (W211901, Aldrich, Germany).
Essential oil composition
A gas chromatograph coupled to a mass spectrometer (QP2010-Plus, Shimadzu, Canby, OR, USA) operated in SCAN mode was employed for analysis of essential oils. Diluted samples (1:10 in n-hexane) of 1 μL were injected manually in the injector at 250°C. Split ratio was 70:1. The capillary column was the Zebron ZB-5MS (30m×0.25mm, 0.25μm film thickness) (Phenomenex, Torrance, California, USA). The temperature program was 40°C for 6 min, then to 160°C at 3°C/min, then to 250°C at 10°C/min, and hold for 10 min. Carrier gas was helium at a constant flow of 1.0 mL/min. The interface was kept at 270°C and the ion source at 250°C. The MS scan parameters included a mass range of 40–400 m/z, a scan speed of 1000 amu/s and a detector voltage of 1.5 kV. Identification was made by combining comparison of the obtained mass spectra with those stored in NIST08 and Wiley7 libraries and retention indices matching. The retention indices were determined in relation to a homologous series of n-alkanes (C8–C20) under the same operating conditions A positive match required both a spectral fit score of ≥95% and a retention index ±5 units of reported values.34 Relative percentages of components were calculated based on GC peak areas.
Preparation of antimicrobial treatment solutions
Sterile solutions of lavender, mint, basil and sage essential oils were prepared in sterile water at 2 different concentrations (Low:100ppm and High:200ppm). The working solutions were diluted in sterile water and treated with ultrasound for 3 minutes, so as to obtain a well-mixed solution. For the Ultrasound treatment, a 5.75 L ultrasound tank (Elmasonic, Germany) was filled with 3 L of distilled water and used at an operating frequency of 37 kHz and a power up to 30 Watts/L. These essential oil treatment solutions were prepared fresh the day of the experiment and used immediately.
Disinfection treatments
EOs treatment
The different essential oil solutions (1mL EO solution) were sprayed separately on, prior inoculated with bacteria or viruses, lettuce surface samples and the samples were kept with EOs solutions for a contact period of 15 min. Then, the samples were washed with sterile water and left to dry in a sterile drying paper (Fisherbrand™ Fluted Qualitative Filter Paper Circles, Thermofischer Scientific, Greece). Control samples followed the same method sprayed with deionized water (1mL deionized water).
US treatment followed by EOs
For US treatment a 5,75 L ultrasound bath (Elmasonic, Germany) was filled with water and used at an operating frequency of 37 kHz and a power up to 30W/L. The lettuce leaves were placed into an empty sterilized beaker, filling it with 300 mL of sterilized water. Ultrasound treatment was applied with a continuous mode for 30 minutes, as this was found in previous study to be the most promising treatment time (Birmpa et al., 2013). After that, the lettuce leaves were left to dry at a sterile drying paper and then the EOs treatment followed. Each experiment was performed with two concentrations of each essential oil (low and high concentration) for a total time of 45 minutes (30 minutes ultrasound followed by 15 minutes of EOs spraying). At least three replicates of each treatment were performed.
UV treatment followed by EOs
For UV treatment, a UV cabinet with four UV-C (Osram Germicidal G5) lamps was used .32 The treatment was conducted at an intensity of 2 mW/cm2 at dosage 3.6 J/cm2. After that the EOs treatment followed. Each experiment was performed with two concentrations of each essential oil (low and high concentration) for a total time of 45 minutes (30 minutes UV followed by 15 minutes of EOs spraying). At least three replicates of each treatment were performed.
Bacterial Enumeration
For enumeration of bacteria, 10 g of treated lettuce sample was transferred into a sterile stomacher bag (Gosselin SM2B-01, Villeurbanne, France) containing 90 mL of Peptone Buffer water (PBW; Oxoid, U.K.) and homogenized in a stomacher (BagMixer, Interscience, St Nom la Bretêche, France) for 2 min, and then all samples were analyzed using ISO enumeration methods according to Birmpa et al.32 Log10 Reductions of bacteria were calculated on a per gram of vegetable basis.
Recovery of viruses from fresh RTE samples and Evaluation of hAdV35 infectivity
RTE samples were processed by the method of Dubois et al.35 with slight modifications, as described by Kokkinos et al.36 and Birmpa et al.37 For the evaluation of the infectivity of hAdV35 all the steps were followed according to Birmpa et al.37
DNase Treatment and Nucleic Acid Extraction
An enzymatic digestion treatment was applied in this study to reduce false positive results by detection of free DNA when using qPCR analysis. Each analyzed sample was treated with DNase I (free – RNase) (DNase I, Molecular Grade, Invitrogen, USA), before DNA extraction to degrade DNA released from damaged viral capsids, according to manufacturer's instructions. Nucleic acids (NA) from viral concentrates derived from fresh produce samples were extracted using a NucliSENS miniMAG kit (bioMerieux, France), according to previous published protocols. A negative control was included in all nucleic acid extraction procedures. Finally, the NA eluates (100μl) were stored at −70°C, until used.
Quantitative real-time PCR (QPCR)
The virus assay was performed using the conditions (primers, probes and amplification conditions) presented by Hernroth et al.38 The initial and final hAdV concentrations were quantified by qPCR. In all the cases, a neat and a 10-fold dilution of the virus nucleic acid extract were tested; all samples were tested in duplicate (two neat and two diluted). The qPCR assays were performed using TaqMan Universal PCR Master Mix (Applied Biosystem, USA) and a carry-over contamination prevention system, uracil N-glycosylase. In each assay, 10μl sample of nucleic acid extract was added, to make a final reaction volume of 25μl. For each plate, the genomic equivalence (GE/ml) was measured. Ultra-pure water was used as the non-template control for each assay.
Statistical Analysis
All experiments were carried out in triplicates. The microbiological data were analyzed in terms of log (N/N0), where N is the microorganism load at a given time, and N0 corresponds to the initial microbial load of untreated samples. The data were analyzed for statistical significance using SPSS 20.0 (SPSS Inc., Chicago, USA). Results were compared by an analysis of variance followed by Tukey's method with a significance level of p<0.05.
Results and discussion
Composition of Essential Oils
The main components of crude essential oils of lavender, salvia, mint and basil are presented in Table 1. In total, 67 - 77 compounds were identified in each EO of the four herbs. Among them, two or three comprise the 20–70% of the total and are the ones that determine the biological properties of the EOs, which is accordance with Bakkali et al.8 In Table 1 only the compounds found in any of the four EOs at percentages >1% are shown. It is observed that linalyl acetate, linalool, camphor and 1,8-cineole are the main components of lavender accounting for almost 70% of its composition, in accordance with Shellie et al.39 The composition of Salvia confirms its chemotype European S. fruticosa, since thujone and 1,8-cineole are the dominant compounds.34 Menthol and menthone are the major compounds in Mentha piperita essential oil in accordance with literature.8 Finally, the commercial basil EO is composed mainly of methyl chavicol (82.4%).
Compound |
Calc. RI |
Ref. RIa |
Lavender |
Salvia (sage) |
Mint | Basil (Sigma) |
|---|---|---|---|---|---|---|
α-pinene |
931 |
937 |
|
2.90 |
|
|
Camphene |
948 |
952 |
|
2.80 |
|
|
β-pinene |
974 |
979 |
|
4.82 |
1.07 |
|
Myrcene |
988 |
991 |
|
4.27 |
|
|
Limonene |
1026 |
1030 |
|
1.94 |
6.09 |
|
Eucalyptol (1,8-Cineole) |
1029 |
1032 |
15.04 |
28.17 |
4.92 |
4.01 |
γ-terpinene |
1056 |
1060 |
|
2.36 |
|
|
cis-linalool oxide |
1069 |
1074 |
3.38 |
|
|
|
trans-linalool oxide |
1084 |
1087 |
2.38 |
|
|
|
Linalool |
1097 |
1099 |
16.09 |
|
|
|
β-Thujone |
1116 |
1114 |
|
17.41 |
|
|
Camphor |
1145 |
1144 |
17.19 |
9.72 |
|
|
Menthone |
1156 |
1154 |
|
|
31.58 |
|
Isomenthone |
1163 |
1164 |
|
|
2.87 |
|
Neo-menthol |
1168 |
1166 |
|
|
3.80 |
|
Borneol L |
1169 |
1166 |
3.86 |
|
|
|
Terpinen-4-ol |
1177 |
1177 |
3.89 |
|
|
|
Menthol |
1178 |
1174 |
|
|
29.09 |
|
Estragole (methyl chavicol) |
1199 |
1196 |
|
|
|
82.41 |
Linalyl acetate |
1252 |
1257 |
21.37 |
|
|
|
Menthol acetate |
1290 |
1294 |
|
|
7.17 |
|
α-terpinyl acetate |
1345 |
1350 |
|
6.55 |
|
|
trans-Caryophyllene |
1414 |
1419 |
1.14 |
5.03 |
|
|
α-trans-bergamotene |
1428 |
1436 |
|
|
|
3.37 |
Table 1 Major components of studied essential oils (in % abundance)
a From: Babushok et al.38
Cytotoxic properties have been attributed to many of the compounds in the EOs. The antibacterial potential of essential oils’ components has been reported to be ranked as follows: Linalyl acetate < limonene < β-pinene < α-pinene < camphor < linalool < 1,8-cineole < menthol < thymol < carvacrol.41 Consequently, the antimicrobial activity of each tested EO is expected to depend on the relative occurrence of its individual constituents. In general, cytotoxicity of EOs is mostly due to alcohols, phenols and aldehydes.8
Effect of use Different Essential Oils (EOs) treatments as disinfectants on fresh RTE lettuce
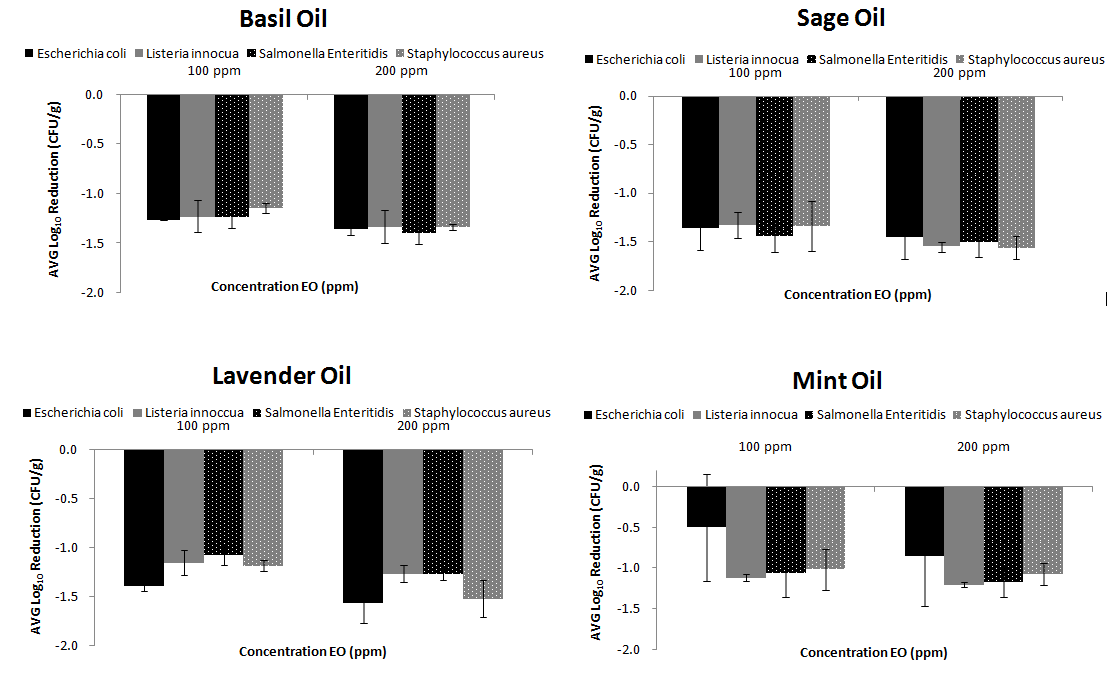
The effect of all EOs used in this study on the survival of E.coli, L. innocua, S. Enteritidis and S. aureus in lettuce’s surfaces are given in Figure 1. The factor “treatment time” was selected to be 15 minutes as it has been shown from other studies30,42 that it is an adequate disinfection time. Gündüz et al.11 studied different washing treatments (5, 10, 15 and 20 min) for disinfecting Salmonella on fresh produce with myrtle oil. They concluded that there was no significant difference between treatment times in reduction of S. Typhimurium.11
|
Figure 1 Disinfection Efficiency of EOs (low and high concentration) for four bacteria prior inoculated on fresh RTE lettuce |
All the EOs concentrations used in this study reduced significantly (p<0.05) the levels of the tested bacteria found on the surface of tested lettuce samples. The best disinfection was achieved with sage EO where log reductions of -1.366±0.22, -1.329±0.14, -1.441±0.16, -1.336±0.26 logs10 were achieved for E.coli, L. innocua, S. Enteritidis and S. aureus respectively, when 100ppm concentration was used. While using 200ppm EO concentration caused reduction -1,456±0.22, -1,551±.0.05, -1,507±0.14, -1,559±0.12 logs10 respectively. (Figure 1). Then basil, lavender and mint EOs followed as far as their disinfection efficiency is concerned.
When the concentration was enhanced from 100 to 200ppm log reductions were observed. For example, log reductions up to 0.4 log10 were observed for all tested microorganisms when the EO concentration was increased.
No significant differences (p>0.05) were shown between different microorganisms when the same EO was implemented on lettuce surfaces. For E.coli, the disinfection was better when lavender and sage EOs were used (-1.559±0.06 logs and -1.456±014 logs respectively) rather than when basil and mint EOs were implemented (-1.357±0.11 logs and 0.857±0.60 logs respectively). Ousallah et al.43 reported that several essential oils had significant inhibitory effect on the survival of E. coli O157:H7 (Karagözlü et al., 2011). Singh et al.44 reported that mint essential oil had an antimicrobial affect against E. coli O157:H7 in lettuce samples. Researchers indicated that with the increasing concentration of the essential oil its antimicrobial affect against pathogen bacteria increased too.42 The data obtained in this study also supported the above findings. The results achieved with basil EO were similar for all microorganisms (p<0.05).
According to our findings, basil EO has mainly estragol in its chemical composition. It is thought that antimicrobial effect of this essential oil is due to this compound present in the composition.42 The best results of Basil EO were observed for S. Enteritidis (-1.389±0.11 log10). Moreover, using mint essential oils at different concentrations had significant effect on the changes in S. Enteritidis counts of the samples (p<0.05), which has also been found in another study for S. Typhimurium.30
Combined treatment of US followed by EOs for disinfection of fresh RTE lettuce
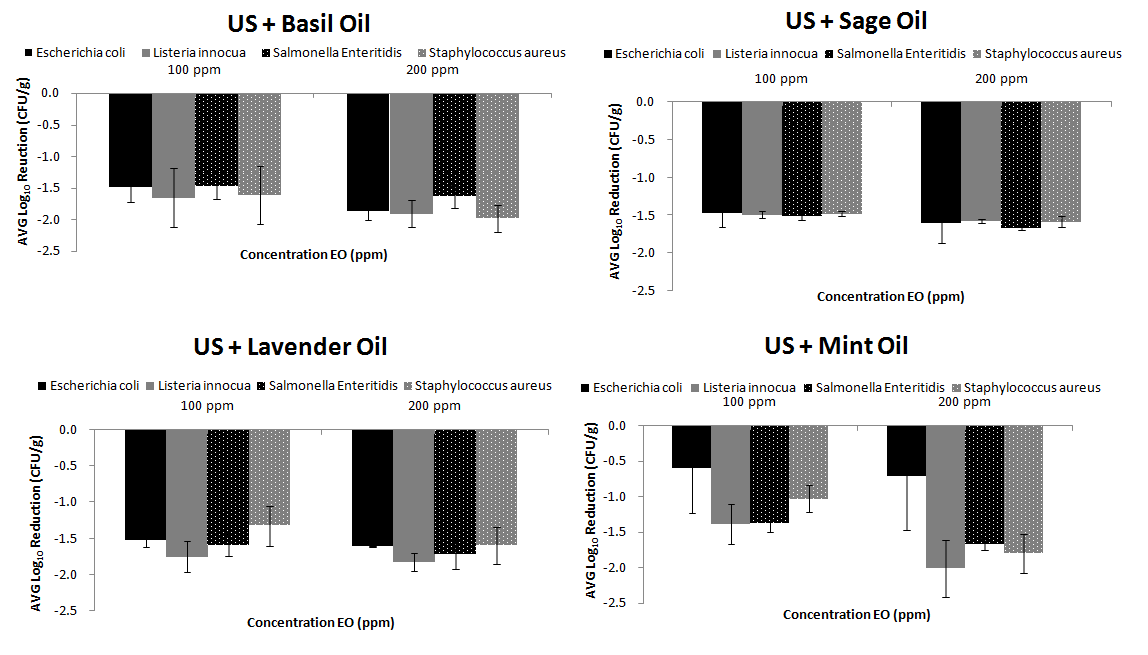
US has been applied on fresh produce to inactivate microorganisms. In our previous study,32 we reported a reduction of 2.30 log10 CFU/g of E. coli on lettuce after 30 min of sonication. Seymour et al.29 applied an US treatment of 70 kHz for 10 min on lettuce leaves and inactivated E. coli at 0.97 log10 CFU/g.
In our study the highest reductions were achieved when US was followed by basil or mint EOs. For example, reductions of -1.982±0.21 and -2±0.40 log10 CFU/g were achieved for S. aureus and L. innocua when US was followed by basil and mint EOs respectively. Then reductions of S. Enteritidis and L. innocua of -1.671±0.03 and -1.823±0.12 log10 CFU/g achieved when US was followed by sage and lavender EOs respectively. No significant reductions (p>0.05) were found when all EOs were combined with US. However, significant reductions existed (p<0.05) for all US/EOs, compared to EOs alone and UV/EOs combinations (Figure 2). In the study of Millan-Sango et al.30 synergistic effect of US combined with 0.025% (v/v) of EO of oregano, for disinfection of E.coli on lettuce leaves was observed.
|
Figure 2 Disinfection Efficiency of Ultrasound disinfection treatment followed by EOs treatment (low and high concentration) for four bacteria prior inoculated on fresh RTE lettuce |
Combined treatment of UV followed by EOs for disinfection of fresh RTE lettuce
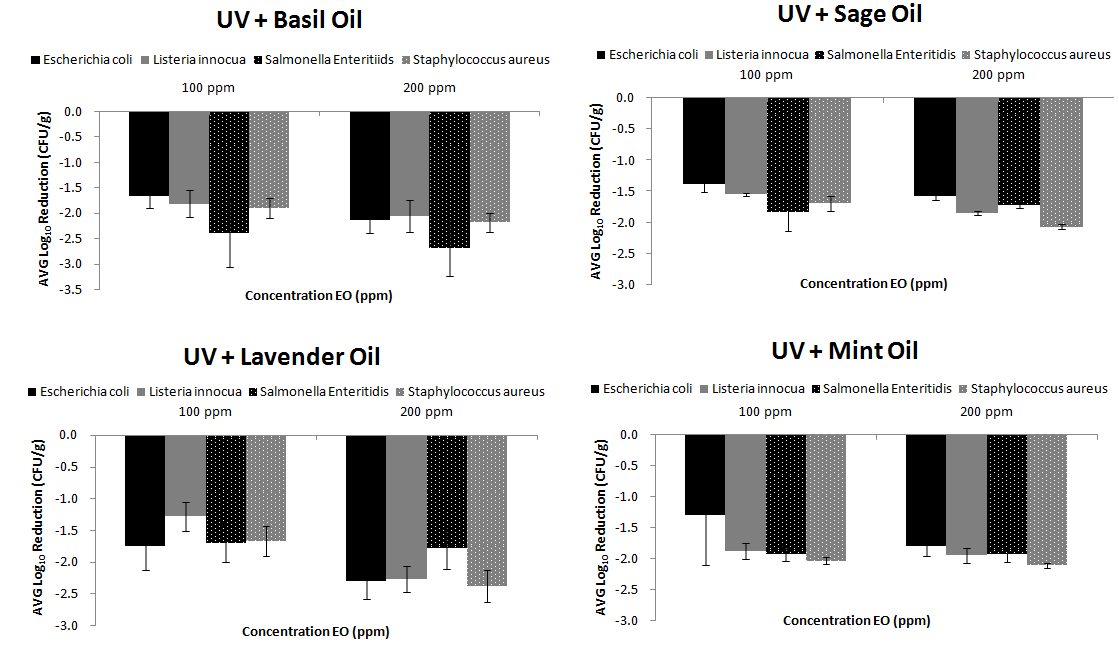
UV disinfection has been applied in our previous study and has significantly reduced populations of E.coli, L. innocua, S. Enteridis, S. aureus in fresh RTE produce.32 The results achieved in the present study when combined UV/EOs were used, enhanced the disinfection efficiency compared to EOs alone for all microorganisms (p<0.05) and US/EOs treatments (p<0.05). For instance, a significant difference was observed between US/Basil EO and UV/Basil EO (p=0.022) as well as between US/Mint EO and UV/Mint EO (p=0.001). However, the difference between US and different EOs was not significant (p>0.05) (Figure 3).
|
Figure 3 Disinfection Efficiency of UV-C disinfection treatment followed by EOs treatment (low and high concentration) for four bacteria prior inoculated on fresh RTE lettuce |
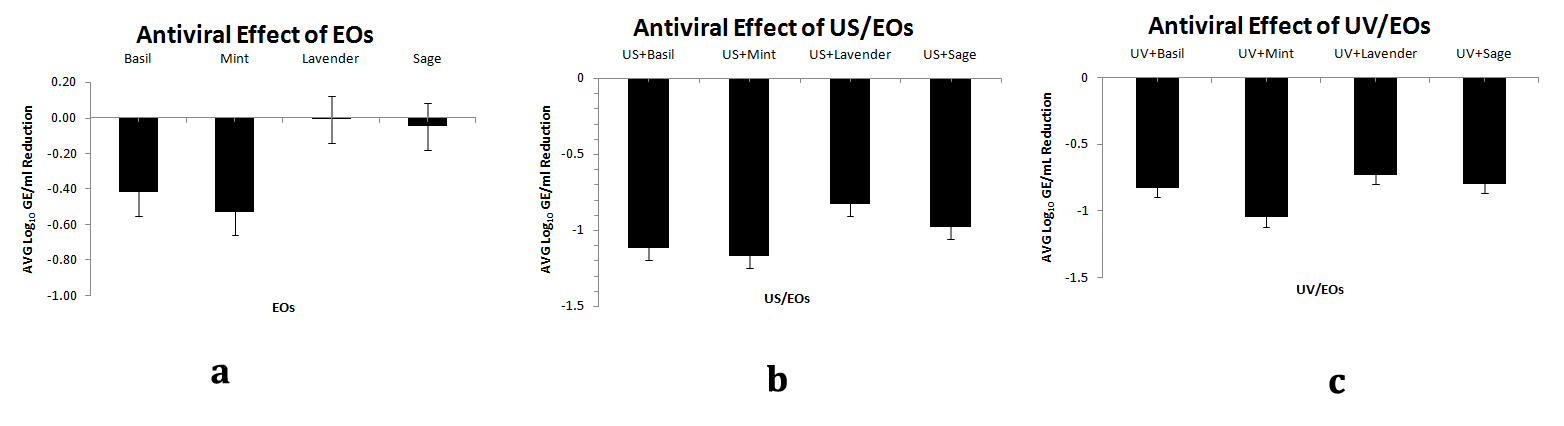
Antiviral Effect of EOs alone or combined with US and UV
The results obtained by sage and lavender EOs did not affect hAdVs concentration (Figure 4A). However, basil and mint EOs exhibited good disinfection results, reaching about 0.60 log10 GE/mL reductions of AdV. Other studies, have studied the antiviral effect of Melaleuca alternifolia in different viruses including adenovirus-2, but no effect was obvious.45 Kovac et al.46 showed that Hyssopus officinalis and Thymus mastichina EOs in concentration of 0.02% had no effect in the reduction of murine norovirus (MNV-1) and human adenovirus (HAdV-2). According to Cermelli et al.47 eucalyptus EO in concentration of 0.25 μl/mL did not also reduce significantly AdV. Su and d’Souza48 studied the effect of GSE against foodborne viral surrogates in lettuce. Their study evaluated the application of GSE (commercial Gravinol-S) against many viruses which involved also hepatitis A virus (HAV) and human norovirus surrogates, feline calicivirus (FCV-F9) and murine norovirus (MNV-1). They also concluded that GSE shows potential for foodborne viral reduction on produce as part of hurdle technologies.
As far as non-thermal disinfection technologies is concerned, existing studies show the effect of UV or US on viruses. Meng and Gerba49 found a 3 log inactivation of adenovirus type 40 (AdV40) at a UV dose of 90 mJ/cm2 and a 4 log reduction at 120 mJ/cm2. Whereas, Thurston-Enriquez et al.50 found that AdV 40 requires over 150 mJ/cm2 for 3 log and over 200 mJ/cm2 for 4 log inactivation. AdV1, AdV2, and AdV6 require 120 mJ/cm2 for 3 log inactivation.51 Chrysikopoulos et al.52 showed that the bacteriophages X174 and MS2, which were used as model viruses, were removed adequately when relatively high US frequencies (i.e., 582, 862, and 1142 kHz) were used.
However, to the author’s knowledge no studies exist which combine EOs with non-thermal technologies, as far as virus disinfection is concerned. In our study the results with the non-thermal technologies (UV and US) enhanced the disinfection ability of EOs and the best results were achieved with Basil and Mint EOs followed by UV and US, compared to the rest EOs (Figures 4B and C).
Conclusion
Consumers demands are in favor of using natural antimicrobials for food disinfection, such as essential oils (EOs) and non-thermal disinfection technologies. The results of the present study showed that differences in disinfection efficiency (p<0.05) among different EOs were obvious. The more efficient disinfectants were mint and basil EOs. Moreover, the combinations of US/EO’s or UV /EO’s were more effective (1-2 log10 reduction) than the EO’s alone (0.5-1 log10 reduction) for reducing microorganisms in fresh RTE lettuce. These data suggest that the combination of non-thermal with EO’s could find potential applications for decontamination of bacteria and viruses in the food industry.
Highlights
- Foodborne pathogens are major contaminants of fresh ready-to-eat foods.
- Basil and mint essential oils are effective against foodborne pathogens.
- Combined essential oils with non-thermal technologies disinfect lettuce.
References
- Slavin JL, Lloyd B. Health benefits of fruits and vegetables. Adv Nutr. 2012;3(4):506–516.
- Ragaert P, Verbeke W, Devlieghere F, Debevere J. Consumer perception and choice of minimally processed vegetables and packaged fruits. Food Qual Prefer. 2004;15(3):259–270.
- Dhandevi PEM, Jeewon R. Fruit and vegetable intake: Benefits and progress of nutrition education interventions-narrative review article. Iran J Public Health. 2015;44(10):1309.
- Olaimat AN, Holley RA. Factors influencing the microbial safety of fresh produce: a review. Food Microbiol. 2012;32(1):1–19.
- Abadias M, Alegre I, Usall J, Torres R, Viñas I. Evaluation of alternative sanitizers to chlorine disinfection for reducing foodborne pathogens in fresh-cut apple. Postharvest Biol Technol. 2011;59(3):289–297.
- Xu W, Qu W, Huang K, et al. Antibacterial effect of grapefruit seed extract on food-borne pathogens and its application in the preservation of minimally processed vegetables. Postharvest Biol Technol. 2007;45(1):126–133.
- Burt S. Essential oils: their antibacterial properties and potential applications in foods—a review. Int J Food Microbiol. 2004;94(3):223–253.
- Bakkali F, Averbeck S, Averbeck D, Idaomar M. Biological effects of essential oils–a review. Food Chem Toxicol. 2008;46(2):446–475.
- Holley RA, Patel D. Improvement in shelf-life and safety of perishable foods by plant essential oils and smoke antimicrobials. Food Microbiol. 2005;22(4):273–292.
- Al Nomaani RSS, Hossain MA, Weli AM, Al-Riyami Q, Al-Sabahi JN. Chemical composition of essential oils and in vitro antioxidant activity of fresh and dry leaves crude extracts of medicinal plant of Lactuca sativa L. native to Sultanate of Oman. Asian Pac J Trop Biomed. 2013;3(5):353–357.
- Gündüz GT, Gönül ŞA, Karapınar M. Efficacy of oregano oil in the inactivation of Salmonella typhimurium on lettuce. Food Control. 2010;21(4):513–517.
- Gutierrez J, Bourke P, Lonchamp J, Barry-Ryan C. Impact of plant essential oils on microbiological, organoleptic and quality markers of minimally processed vegetables. Innov Food Sci Emerg Technol. 2009;10(2):195–202.
- Lanciotti R, Gianotti A, Patrignani F, Belletti N, Guerzoni ME, Gardini F. Use of natural aroma compounds to improve shelf-life and safety of minimally processed fruits. Trends Food Sci Technol. 2004;15(3-4):201–208.
- De Azeredo GA, Stamford TLM, Nunes PC, Neto NJG, De Oliveira MEG, De Souza EL. Combined application of essential oils from Origanum vulgare L. and Rosmarinus officinalis L. to inhibit bacteria and autochthonous microflora associated with minimally processed vegetables. Food Res Int. 2011;44(5):1541–1548.
- Gutierrez J, Barry-Ryan C, Bourke P. Antimicrobial activity of plant essential oils using food model media: efficacy, synergistic potential and interactions with food components. Food Microbiol. 2009;26(2):142–150.
- Siroli L, Patrignani F, Serrazanetti DI, et al. Lactic acid bacteria and natural antimicrobials to improve the safety and shelf-life of minimally processed sliced apples and lamb’s lettuce. Food Microbiol. 2015;47:74–84.
- Bouaziz M, Yangui T, Sayadi S, Dhouib A. Disinfectant properties of essential oils from Salvia officinalis L. cultivated in Tunisia. Food Chem Toxicol. 2009;47(11):2755–2760.
- Loizzo MR, Tundis R, Menichini F, Saab AM, Statti GA, Menichini F. Cytotoxic activity of essential oils from Labiatae and Lauraceae families against in vitro human tumor models. Anticancer Res. 2007;27(5A):3293–3299.
- Fellah S, Diouf PN, Petrissans M, Perrin D, Romdhane M, Abderrabba M. Chemical composition and antioxidant properties of Salvia officinalis L. oil from two culture sites in Tunisia. J Essent Oil Res. 2006;18(5):553–556.
- Hayouni EA, Chraief I, Abedrabba M, et al. Tunisian Salvia officinalis L. and Schinus molle L. essential oils: Their chemical compositions and their preservative effects against Salmonella inoculated in minced beef meat. Int J Food Microbiol. 2008;125(3):242–251.
- Sajadi S. Analysis of the essential oils of two cultivated basil (Ocimum basilicum L.) from Iran. 2006.
- Lee S-J, Umano K, Shibamoto T, Lee K-G. Identification of volatile components in basil (Ocimum basilicum L.) and thyme leaves (Thymus vulgaris L.) and their antioxidant properties. Food Chem. 2005;91(1):131–137.
- Politeo O, Jukic M, Milos M. Chemical composition and antioxidant capacity of free volatile aglycones from basil (Ocimum basilicum L.) compared with its essential oil. Food Chem. 2007;101(1):379–385.
- Suppakul P, Miltz J, Sonneveld K, Bigger SW. Antimicrobial properties of basil and its possible application in food packaging. J Agric Food Chem. 2003;51(11):3197–3207.
- Effati-Daryani F, Mohammad-Alizadeh-Charandabi S, Mirghafourvand M, Taghizadeh M, Mohammadi A. Effect of lavender cream with or without foot-bath on anxiety, stress and depression in pregnancy: a randomized placebo-controlled trial. J Caring Sci. 2015;4(1):63.
- Inouye S, Takizawa T, Yamaguchi H. Antibacterial activity of essential oils and their major constituents against respiratory tract pathogens by gaseous contact. J Antimicrob Chemother. 2001;47(5):565–573.
- D’auria FD, Tecca M, Strippoli V, Salvatore G, Battinelli L, Mazzanti G. Antifungal activity of Lavandula angustifolia essential oil against Candida albicans yeast and mycelial form. Med Mycol. 2005;43(5):391–396.
- Ahmad A, Khan A, Samber N, Manzoor N. Antimicrobial activity of Mentha piperita essential oil in combination with silver ions. Synergy. 2014;1(2):92–98.
- Seymour IJ, Burfoot D, Smith RL, Cox LA, Lockwood A. Ultrasound decontamination of minimally processed fruits and vegetables. Int J Food Sci Technol. 2002;37(5):547–557.
- Millan-Sango D, McElhatton A, Valdramidis VP. Determination of the efficacy of ultrasound in combination with essential oil of oregano for the decontamination of Escherichia coli on inoculated lettuce leaves. Food Res Int. 2015;67:145–154.
- Artés F, Gómez P, Aguayo E, Escalona V, Artés-Hernández F. Sustainable sanitation techniques for keeping quality and safety of fresh-cut plant commodities. Postharvest Biol Technol. 2009;51(3):287–296.
- Birmpa A, Sfika V, Vantarakis A. Ultraviolet light and ultrasound as non-thermal treatments for the inactivation of microorganisms in fresh ready-to-eat foods. Int J Food Microbiol. 2013;167(1):96–102.
- Tawema P, Han J, Vu KD, Salmieri S, Lacroix M. Antimicrobial effects of combined UV-C or gamma radiation with natural antimicrobial formulations against Listeria monocytogenes, Escherichia coli O157: H7, and total yeasts/molds in fresh cut cauliflower. LWT-Food Sci Technol. 2016;65:451–456.
- Baser KHC, Buchbauer G. Handbook of Essential Oils: Science, Technology, and Applications. CRC Press; 2015.
- Dubois E, Hennechart C, Deboosere N, et al. Intra-laboratory validation of a concentration method adapted for the enumeration of infectious F-specific RNA coliphage, enterovirus, and hepatitis A virus from inoculated leaves of salad vegetables. Int J Food Microbiol. 2006;108(2):164–171.
- Kokkinos P, Kozyra I, Lazic S, et al. Harmonised investigation of the occurrence of human enteric viruses in the leafy green vegetable supply chain in three European countries. Food Environ Virol. 2012;4(4):179–191.
- Birmpa A, Bellou M, Kokkinos P, Vantarakis A. Effect of Nonthermal, Conventional, and Combined Disinfection Technologies on the Stability of Human Adenoviruses as Fecal Contaminants on Surfaces of Fresh Ready-to-Eat Products. J Food Prot. 2016;79(3):454–462.
- Hernroth BE, Conden-Hansson A-C, Rehnstam-Holm A-S, Girones R, Allard AK. Environmental factors influencing human viral pathogens and their potential indicator organisms in the blue mussel, Mytilus edulis: the first Scandinavian report. Appl Environ Microbiol. 2002;68(9):4523–4533.
- Shellie R, Mondello L, Marriott P, Dugo G. Characterisation of lavender essential oils by using gas chromatography–mass spectrometry with correlation of linear retention indices and comparison with comprehensive two-dimensional gas chromatography. J Chromatogr A. 2002;970(1-2):225–234.
- Babushok VI, Linstrom PJ, Zenkevich IG. Retention indices for frequently reported compounds of plant essential oils. J Phys Chem Ref Data. 2011;40(4):043101.
- Soković M, Glamočlija J, Marin PD, Brkić D, van Griensven LJ. Antibacterial effects of the essential oils of commonly consumed medicinal herbs using an in vitro model. Molecules. 2010;15(11):7532–7546.
- Karagözlü N, Ergönül B, Özcan D. Determination of antimicrobial effect of mint and basil essential oils on survival of E. coli O157: H7 and S. typhimurium in fresh-cut lettuce and purslane. Food Control. 2011;22(12):1851–1855.
- Oussalah M, Caillet S, Saucier L, Lacroix M. Inhibitory effects of selected plant essential oils on the growth of four pathogenic bacteria: E. coli O157: H7, Salmonella typhimurium, Staphylococcus aureus and Listeria monocytogenes. Food Control. 2007;18(5):414–420.
- Singh N, Singh RK, Bhunia AK, Stroshine RL. Efficacy of chlorine dioxide, ozone, and thyme essential oil or a sequential washing in killing Escherichia coli O157: H7 on lettuce and baby carrots. LWT-Food Sci Technol. 2002;35(8):720–729.
- Garozzo A, Timpanaro R, Bisignano B, Furneri PM, Bisignano G, Castro A. In vitro antiviral activity of Melaleuca alternifolia essential oil. Lett Appl Microbiol. 2009;49(6):806–808.
- Kovač K, Bouwknegt M, Diez-Valcarce M, Raspor P, Hernández M, Rodríguez-Lázaro D. Evaluation of high hydrostatic pressure effect on human adenovirus using molecular methods and cell culture. Int J Food Microbiol. 2012;157(3):368–374.
- Cermelli C, Fabio A, Fabio G, Quaglio P. Effect of eucalyptus essential oil on respiratory bacteria and viruses. Curr Microbiol. 2008;56(1):89–92.
- Su X, D’Souza DH. Grape seed extract for foodborne virus reduction on produce. Food Microbiol. 2013;34(1):1–6.
- Meng QS, Gerba CP. Comparative inactivation of enteric adenoviruses, poliovirus and coliphages by ultraviolet irradiation. Water Res. 1996;30(11):2665–2668.
- Thurston-Enriquez JA, Haas CN, Jacangelo J, Gerba CP. Chlorine inactivation of adenovirus type 40 and feline calicivirus. Appl Environ Microbiol. 2003;69(7):3979–3985.
- Nwachuku N, Gerba CP, Oswald A, Mashadi FD. Comparative inactivation of adenovirus serotypes by UV light disinfection. Appl Environ Microbiol. 2005;71(9):5633–5636.
- Chrysikopoulos CV, Manariotis ID, Syngouna VI. Virus inactivation by high frequency ultrasound in combination with visible light. Colloids Surf B Biointerfaces. 2013;107:174–179.